New course teaches phlebotomy basics
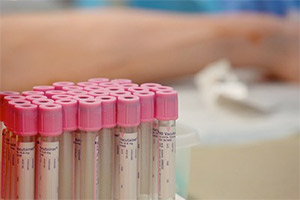
Many clinical research coordinators are called on to step in and draw blood from subjects. But, since there is no requirement that these researchers be trained in phlebotomy, many have no experience. Gait Jordan, MA, clinical research specialist for the Department of Emergency Medicine, has faced this nerve wracking experience himself. So he, along with Valorie Buchholz, RN, BSN, set out to close that skills gap.
“It’s an issue that kept coming up; clinical research coordinators needed these skills, but had never been taught them. We wanted to alleviate those concerns,” Jordan said.
Buchholz – who serves as Associate Director for Clinical Trials Quality Assurance – put together an eight-member advisory board and they began mapping out the content for a course teaching phlebotomy basics to clinical research staff. That course just finished its first session and was, Buchholz said, very well received.
The course “Phlebotomy Training for Clinical Research Staff” covers the basics of drawing blood for research coordinators faced with this responsibility, but with little to no experience. The course covers the process for safely drawing blood, the necessary supplies, how to use them and how to prevent and manage potential complications.
It is open to all School of Medicine employees, but, was structured specifically to cover the skills needed by clinical research staff.
“We are focusing on very basic things: safety protocols, equipment, selecting the right tubes and needles,” Buchholz said.
Classes meet weekly, in one hour sessions, for a total of four weeks. Instruction is split between classroom time and practice in the School of Nursing’s simulation lab. Students must complete a written test with a score of 85 percent or better to move on to the simulation lab where they practice on both artificial arms and human subjects, under the guidance of nurses who volunteer as overseers and instructors.
“We let them practice on each other, then after a while, we even let them practice on us,” Buchholz said.
Buchholz emphasized that this course just covers the basics of phlebotomy; students who complete it are not qualified as Certified Phlebotomists.
Jordan said part of his motivation for putting this course together was the fact that the phlebotomy certification courses that are available are costly, time consuming and go well beyond the knowledge that clinical research staff needs to do what is required of them.
“The purpose of this class is for clinical research coordinators to gain skills. Completing this course just gives them confidence so that the next time they are expected to draw blood they feel comfortable doing so,” Jordan said.
A pilot course was offered earlier this year to five students. Feedback from those initial students helped shape the course content. The first official cohort of 12 students just finished the first training, and Buchholz expects to add more sessions in the coming months, with a goal of offering the classes quarterly.
The program is the result of collaboration among UNC’s Office of Clinical Trials, the Network for Clinical Research Professionals (NCRP), Education-Innovation-Simulation Learning Environment (EISLE) at the School of Nursing, NC TraCS Institute, Health Sciences Library and Andy Johns, Associate Vice Chancellor for Research.
For more information on the course and future sessions, click here.
Course Advisory Board Members:
Gait Jordan, MA, Aylin Regulski, MS, JD, Claudia Christy, RN, Fran Allegri, Juanita Cuffee, MS, Karen Dorman, RN, Emily Olsson and Lauren Bethune
Originally published in
- Created on .